Consulting/Instruction
At KCL, we offer the following diagnosis and instruction services to testing laboratories (ISO / IEC 17025), inspection agencies (ISO / IEC 17020), standard production organizations (ISO guide 34/35) and medical testing laboratories (ISO 15189) for accreditation of public organizations.

Scope of Work
- Establishing a quality and technical system based on international standards, regulations related to the KOLAS compliance, and the characteristics of individual agencies
- Instruction on the established quality management system
- Instruction on the estimation of the uncertainty of measurement (by category)
- Instruction on statistical analysis in relation to the operation of test quality system

Accreditation Procedure & Instruction Details
- Organizations preparing for the initial accreditation: 10 ~ 14 months
- Organizations re-establishing their management system : 2 ~ 4 months
- Organizations adding new areas of accreditation : 4 ~ 8 months
Accreditation Procedure & Instruction Details
- Organizations preparing for the initial accreditation: 10 ~ 14 months
- Organizations re-establishing their management system : 2 ~ 4 months
- Organizations adding new areas of accreditation : 4 ~ 8 months
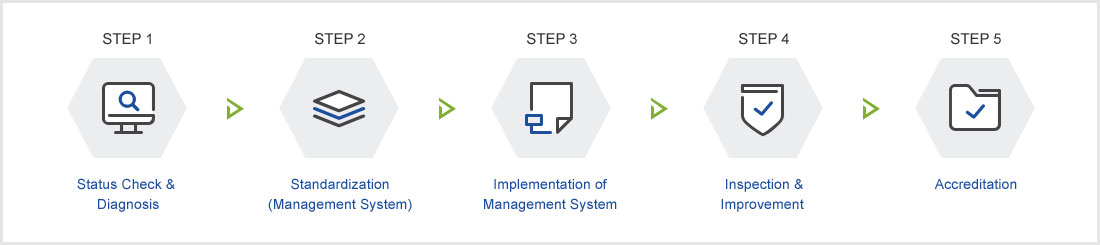
| Step 1. Status Check & Diagnosis |
|
|---|---|
| Step 2. Standardization (Management System) |
|
| Step 3. Implementation of Management System |
|
| Step 4. Inspection & Improvement |
|
| Step 5. Accreditation |
|